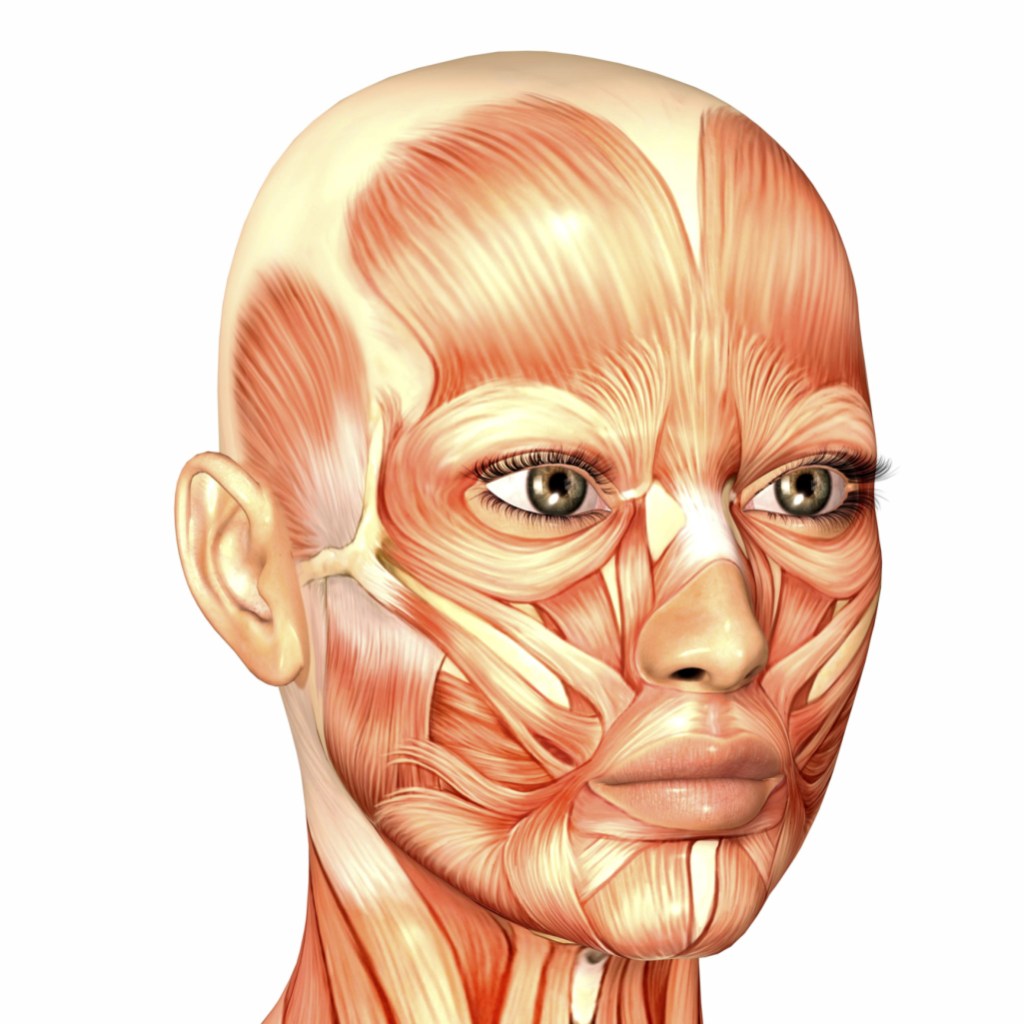

In 1965, while I was a student of Human Anatomy at Kurnool Medical College, I had the opportunity to know about Dr. J. C. B. Grant (1886-1973), the author of Grant’s Atlas of Anatomy. The 5th Edition of his Atlas was published in 1962 and was available in India in our Medical College Library.
Born in Loanhead (south of Edinburgh) in 1886, Grant studied medicine at the University of Edinburgh Medical School and graduated with an M.B., Ch.B. degree in 1908. While at Edinburgh, he worked under the renowned anatomist Daniel John Cunningham. Grant became a decorated serviceman of the Royal Army Medical Corps during the First World War before moving to Canada. He established himself as an ‘anatomist extraordinary’ at the University of Toronto, publishing three textbooks that form the basis of Grant’s Anatomy. The textbooks are still used in anatomy classes today, and made unforgettable memories for those who found themselves in his classes nearly a century ago. One of Grant’s many accomplishments was establishing a division of histology within the department.

As a medical student, I used Grant’s Atlas of Anatomy, the seminal work of Scottish-born Dr. John Charles Boileau Grant, who would become the chair of Anatomy at the University of Toronto in 1930 and retired in 1965.
John Charles Boileau Grant (1886–1973)


The author of Grant’s Atlas of Anatomy (1943), Grant used to train thousands of medical students around the world. He came to University of Toronto’s Faculty of Medicine from University of Manitoba (and previously Edinburgh), and was Chair of the Department of Anatomy there from 1930 to 1965. Although he is best known for this famous atlas, his research and teaching also included biological anthropology, as evidenced by such work as Anthropometry of the Cree and Saulteaux Indians in Northeastern Manitoba (Archaeological Survey of Canada 1929). The human skeletal collection he formed, the “J.C.B. Grant Collection,” is still a core collection for human osteology in the Department of Anthropology at University of Toronto. He is also remembered in the Grant’s Museum at the Medical Sciences Building at the University of Toronto. This museum, with its displays of anatomical specimens, many of which were dissected by Grant himself, continues to be used in an active learning environment by more than 1000 students each year.
Students continue to use Grant’s textbooks today, and for the more artistic anatomist there’s even a Grant’s Anatomy Coloring Book, published in 2018.

At the University of Toronto, Dr.McMurrich, Chair of Anatomy was succeeded as chairman in 1930 by Dr. John Charles Boileau Grant. Dr. Grant wrote three text books, of which “An Atlas of Anatomy” (published in 1943) rapidly gained international prominence and is still, one of the most widely used anatomical atlases in the world. It is now known as “Grant’s Atlas of Anatomy” and is in its tenth edition. The atlas was based on a series of elegant dissections done either by Grant or by others under his supervision. Many of these dissections are currently housed in Grant’s Museum at the University of Toronto.
The Rudi-Grant Connection is about knowing the man, the building blocks and the structural units and organization of the human body. To defend the human existence, the Rudi-Grant Connection lays the emphasis on knowing the person who is at risk apart from knowing the agent posing the risk.
THE IDENTITY OF MULTICELLULAR HUMAN ORGANISM:

Daniel John Cunningham was born on 15 April 1850 in Scotland. After his initial schooling at his home town, Crieff, he took up the study of medicine at the University of Edinburgh and passed with honours. He is best known for the excellent series of dissection manuals, namely Cunningham’s Dissection Manuals.
I learned about the human body while dissecting the body in a systematic manner. The Manual of Practical Anatomy which guides us through this entire process was published in England. The author Dr. Daniel John Cunningham prepared the Manual while dissecting cadavers of British or Irish citizens. He had never encountered cadavers of Indian citizens. At Kurnool Medical College, Kurnool, Andhra Pradesh, India, where I was a student, the Department of Anatomy obtains dead bodies from Government General Hospital Kurnool and most of the deceased are the poor, illiterate, and uneducated people of that region. None of the deceased had the chance to know this man called Cunningham and Cunningham had no knowledge about the existence of these people who arrive on our dissection tables. But, as the dissection of the human body proceeds, inch, by inch, we recognize the anatomical parts as described by Cunningham. The manual also lists some anatomical variations and we very often exchange information between various dissection tables and recognize the variations mentioned. The dissections also involve slicing the organs and studying them, both macroscopically, and microscopically. We did not miss any part of the human body. So what is the Identity of this Human person or Human subject? How does the living Human organism maintain its Identity and Individuality? Apart from the Cultural Traditions of India, several Schools of Religious Thought claim that the Human Individual and its Identity is represented by Human Soul. Where does this soul exist in the human body? What is the location if the soul is present in the living person? Does man have a soul?
The Emotional Experience of Atman as Ananda, Pure Joy, or Pure Bliss:

Atman is a Sanskrit term which describes the spiritual life principle found in all living things, especially regarded as inherent in the real or true Self of the human individual. For all purposes of conversation, Indians use the term Atman to speak about a person’s Soul which is distinct from the Body, and Mind of the person.

Indian thinkers speak extensively describing in great detail the concept of the Atman. Apart from characteristics such as imperishable, indestructible, and immutable, the Atman is viewed as ‘Light’ that dispels the darkness called Ignorance. Indian thinkers adamantly refuse to describe the structural and the functional attributes of the Atman making it difficult to define the term Atman using the information provided by Human Anatomy and Human Physiology. However, there is general agreement among the Indian thinkers about the nature of the Atman. There are four recurrent themes in the discourse about the Atman. These are, 1. The association of the Atman as the ultimate source of Great Knowledge to overcome the veiling effects of Maya or the Grand Illusion, 2. The experience of the Atman is the prerequisite to find Peace, Harmony, and Tranquility in the conditioned nature of the human existence characterized by pain, and suffering, 3. The Atman is manifested as the emotional experience of Ananda, Pure Joy, Perfect Happiness, and Pure Bliss Consciousness, and 4. The Atman is the Fourth Condition, the Fourth State, or the Fourth Quarter of Consciousness which is Pure for it is devoid of all contents, has no functional attributes, and most importantly, cannot be described.

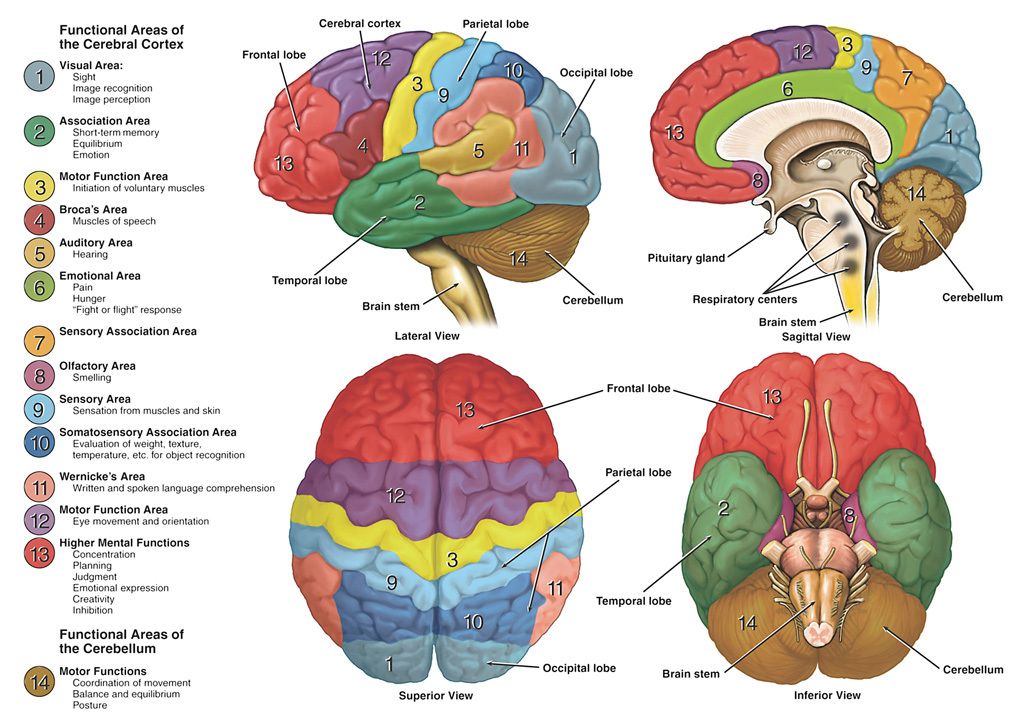
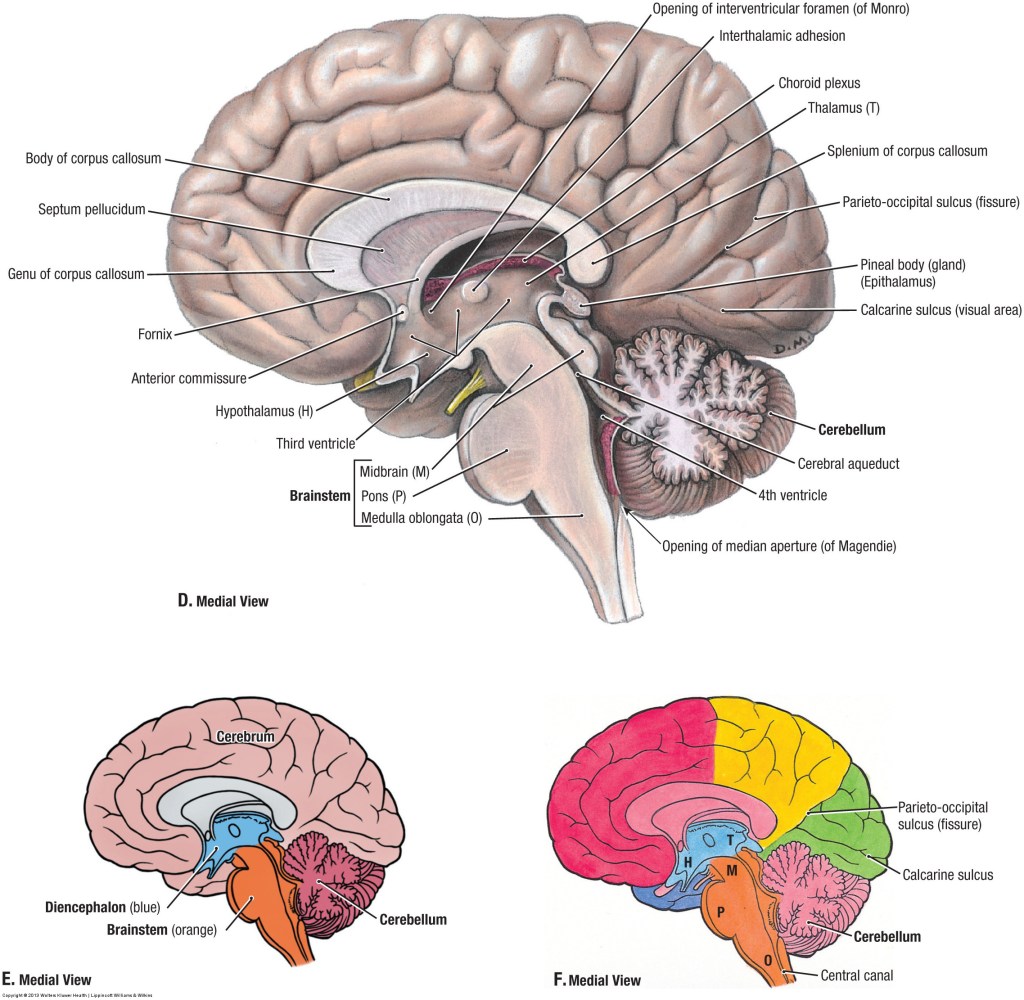
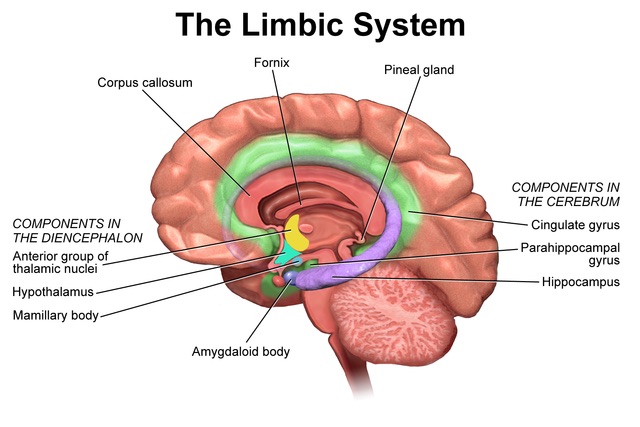
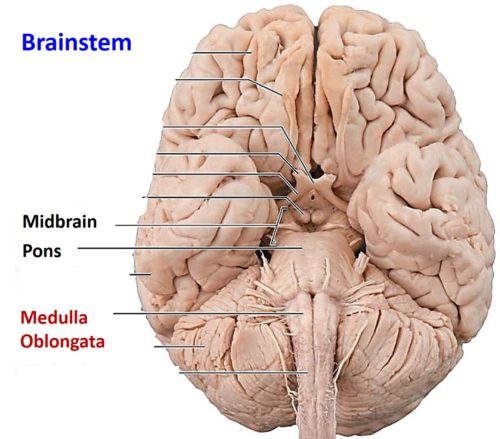
In my analysis, the concept of the Atman, or the Soul is useful and when validated, the concept will provide the tools for practical application to promote the human well-being. To that extent, I invite my readers to study the Functional Anatomy of the Reticular Formation of the Brainstem to interpret it as the structural and functional organization called the Soul. Please review the concept of ‘Emotional Brainstem’ to understand the anatomical and physiological basis of the human emotional experience called Ananda, Pure Joy, Perfect Happiness, and Pure Bliss.
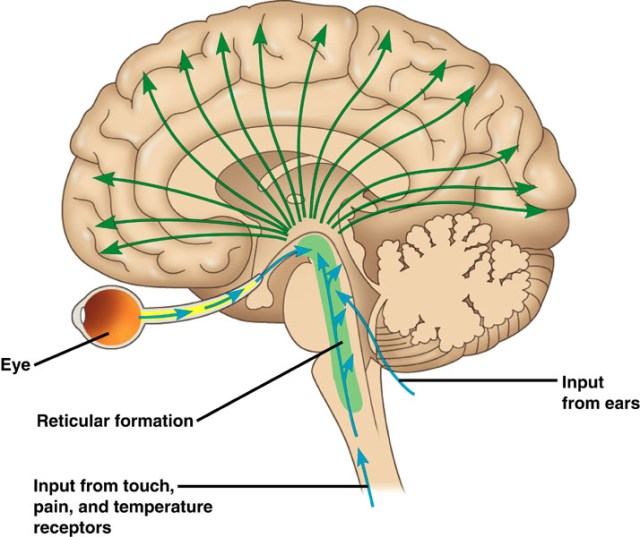
The Functional Anatomy of the Reticular Formation
Frontiers in Neuroanatomy., 29 May 2019 | https://doi.org/10.3389/fnana.2019.00055
Ugo Faraguna1, Michela Ferrucci1,  Filippo S. Giorgi1,2 and Francesco Fornai1,3*
Filippo S. Giorgi1,2 and Francesco Fornai1,3*
- 1Department of Translational Research and New Technologies in Medicine and Surgery, University of Pisa, Pisa, Italy
- 2Section of Neurology, Department of Clinical and Experimental Medicine, Pisa University Hospital, University of Pisa, Pisa, Italy
- 3I.R.C.C.S. I.N.M. Neuromed, Pozzilli, Italy
https://www.frontiersin.org/articles/10.3389/fnana.2019.00055/full
In 1949 Moruzzi and Magoun first described the activating role of a wide area within the brainstem. They defined some physiological features of what they called the ascending reticular activating system, setting the scene for the discovery of the multifaceted roles of the reticular formation. In particular, beyond the original effects on behavioral arousal, a variety of effects is generated in the brain by the activation of these discrete nuclei population of the brainstem. In this way, physiological conditions such as the sleep-waking cycle, the level of arousal and attention, the drive for novelty seeking behaviors, the mood states and other brain activities were shown to depend on the ascending reticular formation. Meanwhile, it became more and more evident that an equal amount of processes is controlled by its descending pathways. More specifically, the reticular formation plays a key role in the modulation of posture, extrapyramidal movements, cardiovascular activity, breathing and a variety of harmonic variations in the sympathetic and parasympathetic systems which accompanies motor activity. The descending fibers of the reticular formation, as well as the ascending system, are critical in gating the sensory inputs and play a critical role in pain modulation, mainly by acting on the posterior horn of the spinal cord.
All these activities are impaired when a damage affects critical nuclei of the reticular formation. This may occur either suddenly, due to vascular disorders, or progressively, as it happens in neurodegenerative conditions. Interestingly, in this latter case the spreading of neurodegeneration has been attributed to the rich collaterals connecting various reticular nuclei, which are more and more involved in later stages of many neurodegenerative disorders.
During the last decades the anatomical counterparts of the reticular formation have been further investigated, even though a comprehensive description is still missing. Thus, the present research topic is designed to welcome contributions both defining the updated anatomy of the reticular formation and its physiological functions (sleep-wake cycle, EEG synchronization, postural control, etc.) as well as its involvement in a wide array of neuropsychiatric disorders (Parkinson and extrapyramidal disorders, epilepsy, sleep disorders, ADHD, degenerative dementia, neurovascular disorders, etc.).
The brainstem reticular formation (RF) represents the archaic core of those pathways connecting the spinal cord and the encephalon. It subserves autonomic, motor, sensory, behavioral, cognitive, and mood-related functions. Its activity extensively modulates cortical excitability, both in physiological conditions (i.e., sleep-wake cycle and arousal) and in disease (i.e., epilepsies). Such a wide variety of effects arises from the long course and profuse axonal branching of isodendritic reticular neurons, which allows the neuronal message to travel toward the entire cerebral cortex and downstream to the spinal cord. On the other hand, the isodendritic architecture featuring a monoplanar branching allows most RF neurons to cover roughly half of the brainstem and to be impinged by ascending and descending pathways. In parallel, such a generalized influence on CNS activity occurs in combination with highly focused tasks, such as those involved in the coordination of gaze.
The Journal Frontiers in Neuroanatomy offers an updated view to define the anatomical correlates of the multiple and interconnected roles played by the brainstem reticular formation in health and disease.
In fact, the integration of multiple activities within the brainstem reticular circuitries may explain why alterations of each of these domains may affect the emotional sphere, paving the way to the concept of emotional brainstem (Venkatraman et al.).
The Brainstem in Emotion: A Review
Anand Venkatraman1,  Brian L. Edlow2 and Mary Helen Immordino-Yang3,4,5*
Brian L. Edlow2 and Mary Helen Immordino-Yang3,4,5*
- 1*. Department of Neurology, University of Alabama at Birmingham, Birmingham, AL, USA
- 2*.Department of Neurology, Massachusetts General Hospital and Harvard Medical School, Boston, MA, USA
- 3*.Brain and Creativity Institute, University of Southern California, Los Angeles, CA, USA
- 4*.Rossier School of Education, University of Southern California, Los Angeles, CA, USA
- 5*.Neuroscience Graduate Program, University of Southern California, Los Angeles, CA, USA
Emotions depend upon the integrated activity of neural networks that modulate arousal, autonomic function, motor control, and somatosensation. Brainstem nodes play critical roles in each of these networks, but prior studies of the neuroanatomic basis of emotion, particularly in the human neuropsychological literature, have mostly focused on the contributions of cortical rather than subcortical structures. Given the size and complexity of brainstem circuits, elucidating their structural and functional properties involves technical challenges. However, recent advances in neuroimaging have begun to accelerate research into the brainstem’s role in emotion. In this review, we provide a conceptual framework for neuroscience, psychology and behavioral science researchers to study brainstem involvement in human emotions. The “emotional brainstem” is comprised of three major networks – Ascending, Descending and Modulatory. The Ascending network is composed chiefly of the spinothalamic tracts and their projections to brainstem nuclei, which transmit sensory information from the body to rostral structures. The Descending motor network is subdivided into medial projections from the reticular formation that modulate the gain of inputs impacting emotional salience, and lateral projections from the periaqueductal gray, hypothalamus and amygdala that activate characteristic emotional behaviors. Finally, the brainstem is home to a group of modulatory neurotransmitter pathways, such as those arising from the raphe nuclei (serotonergic), ventral tegmental area (dopaminergic) and locus coeruleus (noradrenergic), which form a Modulatory network that coordinates interactions between the Ascending and Descending networks. Integration of signaling within these three networks occurs at all levels of the brainstem, with progressively more complex forms of integration occurring in the hypothalamus and thalamus. These intermediary structures, in turn, provide input for the most complex integrations, which occur in the frontal, insular, cingulate and other regions of the cerebral cortex. Phylogenetically older brainstem networks inform the functioning of evolutionarily newer rostral regions, which in turn regulate and modulate the older structures. Via these bidirectional interactions, the human brainstem contributes to the evaluation of sensory information and triggers fixed-action pattern responses that together constitute the finely differentiated spectrum of possible emotions.
Introduction
Emotions are mental and bodily responses that are deployed automatically when an organism recognizes that a situation warrants such a reaction (Damasio, 1994). Due to humans’ intellectual capacities, human emotional reactions are not necessarily triggered by immediate (real) physical or social circumstances, but can also be precipitated by inferences, memories, beliefs or imaginings (Immordino-Yang, 2010). Although human emotions can involve complex cognitive deliberations (Immordino-Yang, 2010, 2015) their activating power fundamentally depends upon the modulation of arousal, motor control and somatosensation. Emotions are therefore regulated by a broad range of subcortical and cortical structures, with a critical role being played by subcortical nuclei in the pontine and midbrain tegmentum (Nauta, 1958; Parvizi and Damasio, 2001), as well as by autonomic and cardiorespiratory nuclei in the medulla (Edlow et al., 2016). Currently, most investigations of human emotion, especially in the neuropsychology literature, have focused on contribution of cortical rather than subcortical structures to human emotion, with a few notable exceptions (Buhle et al., 2013). Given that the brainstem plays a critical role in regulating and organizing emotion-related processing, the aim of this review is to provide a conceptual framework for affective researchers to study the brainstem’s role in human emotion.
Organization of Brain Regions Involved in Emotion
For the purpose of studying its role in emotion, the brainstem can be conceptualized as being composed of Ascending, Descending, and Modulatory networks. The gray matter nodes and white matter connections within each of these networks are summarized in Table 1, while Figure 1 provides a schematic overview of the networks’ brainstem nodes.

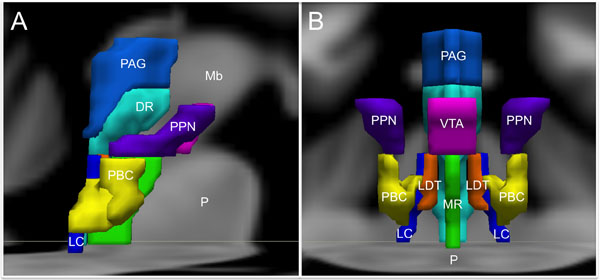
Integration of signaling within these three networks occurs at all levels of the brainstem, while progressively more complex levels of integration occur in the thalamus, hypothalamus and cerebral cortex. This encephalization and hierarchical organization allows phylogenetically older pathways in the brainstem, which evaluate sensory information and give rise to fixed-action pattern responses, to be regulated by evolutionarily newer rostral regions (Tucker et al., 2000). It is important to emphasize here that this conceptual model is based upon limited information about the functioning of the human brainstem, and will likely require revision and further differentiation as new evidence arises (Seeley et al., 2007; Coenen et al., 2011; Hermans et al., 2014).
Ascending Network
Damasio’s (1996) Somatic Markers Hypothesis suggests that emotion processing incorporates somatosensory and visceral feedback from the periphery, either directly or through intervening sensory representations in caudal structures. Multiple representations of the body state in the brainstem and in the insular cortices are believed to enable simulation of future actions and sensations to guide decision making, as well as to contribute to empathy and theory of mind in humans. Self-awareness may arise from successive temporal representations of the body with increasing levels of detail (Craig, 2003a). Even the simple sensory representations of the body in the brainstem nuclei can alter affective experience, as demonstrated by studies showing that subtle modulation of a subject’s facial expressions can change self-reported affect (Harrison et al., 2010).
Interoception, which is the sense of the internal condition of the body, and emotional feeling, may share a common route through the brainstem to the anterior insular cortex (Craig, 2003a; Drake et al., 2010). The interoceptive system, represented in the cortex by the insula and adjacent regions of the frontal operculum, is particularly important for the internal simulation of observed emotion in humans (Preston et al., 2007; Pineda and Hecht, 2009) and for the experience of complex social emotions (Immordino-Yang et al., 2009, 2014, 2016). The other body map in the somatosensory cortex, which is built from dorsal column inputs and segments of the anterolateral pathway, contributes to affective understanding by simulation of facial expressions (Pineda and Hecht, 2009), analogous to the proposed function of primate mirror neurons in perception/action coupling (Rizzolatti and Craighero, 2004).
The neuroanatomic basis for the Ascending sensory network and the mechanisms by which it modulates human emotion remain poorly understood. Although the structural and functional properties of these ascending pathways have been studied extensively in rodents and non-human primates using premortem tract-tracing and invasive electrophysiological studies, these techniques cannot be applied in humans. Recent studies using diffusion tractography and resting-state functional connectivity techniques in humans have found that forebrain regions involved in regulation of mood and affect are interconnected not only with mesencephalic and pontine arousal nuclei, but also with medullary cardiorespiratory and autonomic nuclei through the medial and lateral forebrain bundles (Vertes, 2004; Edlow et al., 2016). Figure 2 provides an overview of the main structures in the Ascending network.
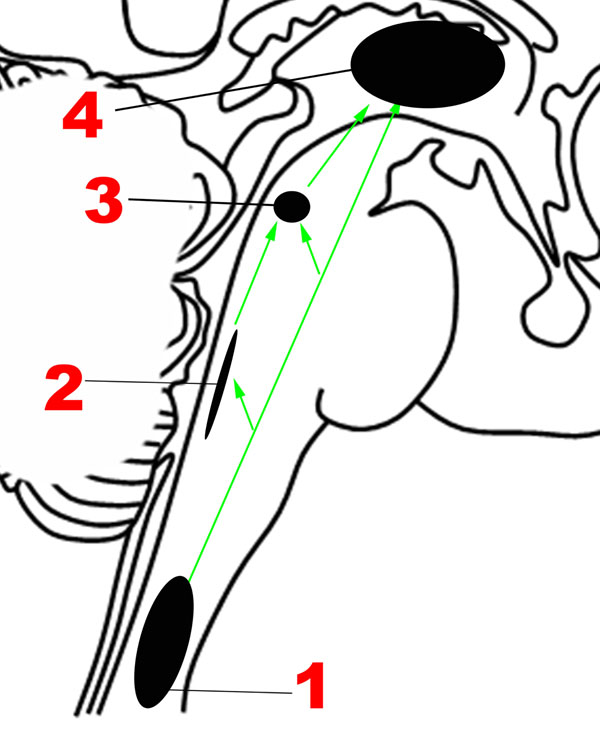
It is well established that sensations from the human body are carried in two major ascending pathways in the brainstem – the dorsal columns of the spinal cord, which continue as the medial lemnisci, carry discriminatory sensation, deep touch and proprioception; the anterolateral pathway, composed of the spinothalamic tracts, carries nociceptive and temperature-related signals (Nogradi et al., 2000-2013).
The Anterolateral Pathway
The nociceptive fibers in the anterolateral pathway give off collaterals at every level that converge with projections from visceral sensory neurons in the brainstem, thereby ensuring close coordination of pain and autonomic processing (Craig, 2003b). The pathway begins with small-diameter fibers that transmit signals of fast and slow pain, chemical changes, temperature, metabolic state of muscles, itch, and sensual or light touch to lamina I of the spinal cord, from where ascending projections arise. In the caudal brainstem, these projections target the nucleus of the tractus solitarius in the medulla (Figure 2), which is also innervated by visceral and taste sensations through the vagus, glossopharyngeal and facial nerves.
The Parabrachial Complex
Tract-tracing studies in rodent models have revealed that ascending projections from the nucleus of the tractus solitarius travel to the parabrachial complex (Figures 1, 2) in the upper pons (Herbert et al., 1990), which also receives direct projections from lamina I neurons (Craig, 2003b), in addition to other inputs such as balance (Balaban, 2002). Rat studies suggest that the parabrachial complex integrates multiple types of converging sensory inputs and in turn projects to rostral regions such as the thalamus, hypothalamus, basal forebrain and amygdala, and may play an important role in arousal (Fuller et al., 2011; Edlow et al., 2012). The upper brainstem, where the parabrachial complex lies, is therefore the most caudal structure where a topographically complete map of the body can be assembled that includes all manner of interoceptive information (Damasio and Carvalho, 2013). There is also ongoing investigation of the role played by the superior colliculus, a structure in the dorsal aspect of the upper brainstem, in sensory and emotional processing in humans, but the available evidence is sparse (Celeghin et al., 2015).
The Thalamus
Immediately rostral to the upper brainstem is the thalamus, and the spinothalamic tracts, as their name indicates, end in the thalamus. A subset of thalamic nuclei function as relay structures between the emotional brainstem and rostral brain structures. The ventral posteromedial nuclei of the thalamus, which receive projections from the parabrachial complex and other parts of the anterolateral pathway, project to the insular cortex, particularly the mid/posterior dorsal part. Craig and colleagues suggested that the posterior part of the ventral medial nucleus of the thalamus, or VMPo, was uniquely involved in pain processing, particularly in primates (Craig, 2003a), but other authors had questioned the separate existence of this nucleus (Willis et al., 2002).
The intralaminar nuclei of the thalamus receive non-topographical sensory input from the spinal cord, which are in turn projected to the orbitofrontal and anterior cingulate cortices. The intralaminar nuclei are involved in orienting and attention, while arousal and visceral sensation are subserved by the midline nuclei (Morgane et al., 2005). In primates a direct pathway from lamina I to the anterior cingulate through the medial dorsal nucleus is also present (Craig, 2003a), and it has been suggested that these pathways may mediate the affective aspect of pain (Tucker et al., 2005). Indeed, the mediodorsal nucleus progressively increases in cytoarchitectonic complexity in higher animals, and is also known to project to the frontal and prefrontal cortices (Morgane et al., 2005). Thus, the thalamus contains multiple structures that appear to play a role in transmitting the signals essential for emotion processing from the brainstem to the forebrain.
Summary statement: Representations of the body of varying degrees of complexity that exist at multiple levels in the Ascending network, including the nucleus of the tractus solitarius and the parabrachial nucleus, are believed to be give rise to the “feeling” of an emotion.
Descending Network
The chief descending pathway in the human brainstem is composed of large, myelinated axons of the corticospinal tracts, transmitting motor impulses to the anterior horn cells of the spinal cord and thereafter to skeletal musculature (Nogradi and Gerta, 2000–2013). In addition, the midbrain and pontine tegmentum, as well as the medulla, contain several structures that serve as the output centers for motor and autonomic regulatory systems, which in turn regulate the bodily manifestations of the “emotion proper” (Damasio, 1994). Holstege (2009) considered the interconnected network of descending fibers and effector regions in the brainstem an “emotional motor system,” distinct from the corticospinal somatic motor pathway, each of which they divided into lateral and medial parts [Figure 3, adapted from (Holstege, 2016)].
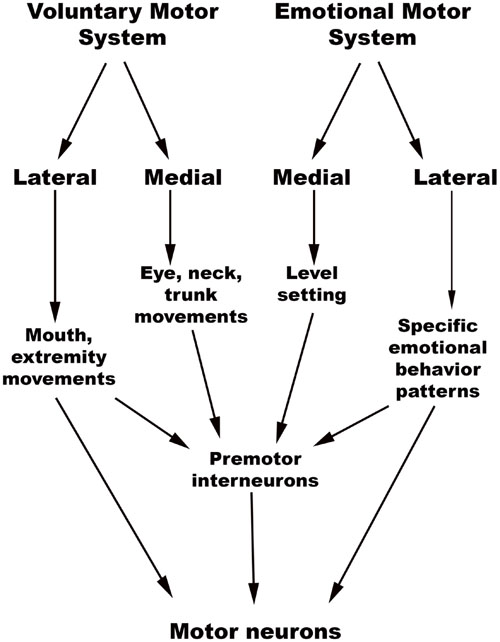
The brainstem, as noted previously, contains a hierarchy of circuits linking ascending sensory neurons and descending effector neurons. Evidence from rat and cat studies indicates that the lower-level circuits enable quick stereotypical responses to stimuli, while the higher-level involvement of rostral centers allows for complex motor and autonomic activity and action specificity (Bandler et al., 2000; Gauriau and Bernard, 2002). This close relationship between sensory and effector networks in emotion processing is best illustrated by the close overlap seen between sites involved in emotional vocalization and pain processing in animals. Both physical and psychological pain (caused by separation from caregivers, for example) can produce distress vocalizations in animals, with the caudal brainstem containing multiple regions that control the respiratory and phonetic changes of vocalization (Tucker et al., 2005) and cardiorespiratory function during emotion (Lovick, 1993; Rainville et al., 2006; Edlow et al., 2016). The rostral nuclei are able to modulate the activity of caudal nuclei that control cardiorespiratory control and vocalization in a coordinated manner that makes the resultant action more complex and nuanced.
Lateral Part of the Emotional Motor System
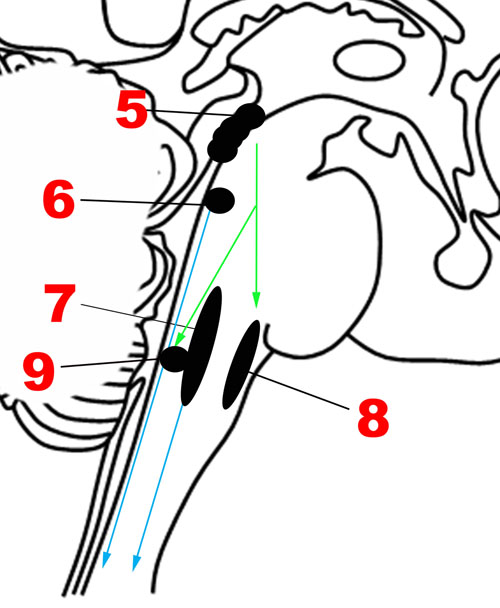
The emotional motor system’s lateral part consists of projections primarily from the periaqueductal gray, as well as more rostral structures such as the amygdala and hypothalamus, to the lateral tegmentum in the caudal pons and medulla (Figures 3, 4). This lateral part of the emotional motor system is involved in specific motor actions invoked in emotions, as well as in the control of heart rate, respiration, vocalization, and mating behavior (Holstege, 2009). Studies in multiple animal models as well as in humans have revealed that the periaqueductal gray (Figures 1, 4) is a major site of integration of affective behavior and autonomic output, with strong connections to other brainstem structures (Behbehani, 1995).
Several fixed patterns of behavior, particularly those related to responding to external threats, with accompanying autonomic changes, are organized in the different columns of the periaqueductal gray in rats (Brandao et al., 2008). The lateral/dorsolateral column receives well-localized nociceptive input (superficial ‘fast’ pain, as might be expected from bites or scratches) and is believed to organize fight-or-flight reactions. When stimulated this column produces emotional vocalization, confrontation, aggression and sympathetic activation, shown by increased blood pressure, heart rate, and respiration. Many of these responses are mediated by descending projections to the paragigantocellularis lateralis nucleus in the rostral ventrolateral medulla (respiratory rhythm), the dorsal motor nucleus of the vagus (heart rate and rhythm), and caudal raphe (cardiorespiratory integration; Lovick, 1993; Edlow et al., 2016). Within this dorsolateral/lateral column itself, there are two parts. The rostral part is responsible for power/dominance (producing a “fight” response), while the caudal part invokes fear (producing a “flight” response) with blood flow to the limbs (Sewards and Sewards, 2002).
The ventrolateral column of the periaqueductal gray receives poorly localized “slow, burning” somatic and visceral pain signals, and on stimulation produces passive coping, long-term sick behavior, freezing with hyporeactivity and an inhibition of sympathetic outflow (Parvizi and Damasio, 2001; Craig, 2003b; Brandao et al., 2005; Benarroch, 2006). In this way, it is likely involved in background emotions such as those that contribute to mood. Rat studies have further revealed that lesions of the dorsolateral periaqueductal gray reduce innate defensive behaviors, while lesions of the caudal ventrolateral part reduce conditioned freezing and increase locomotor activity (Brandao et al., 2005). When the predator is far away, the ventromedial prefrontal cortex and the hippocampus, through the amygdala, activate midbrain structures centered around the ventrolateral periaqueductal gray, which results in freezing (Tucker et al., 2000). In the “circa-strike” stage when the predator is imminent, forebrain pathways are silenced, and the dorsolateral periaqueductal gray is activated, resulting in fight-or-flight reactions.
The Periaqueductal Gray in Human Emotion
Though the reactions detailed above are almost certainly incorporated into human emotion, the precise mechanisms have not been elucidated. One study involving high-resolution MRI of the human periaqueductal gray indicated that this structure has discrete functional subregions that parallel the divisions seen in animals – aversive stimuli caused activation in the ventrolateral regions of the caudal periaqueductal gray and in the lateral/dorsomedial regions of the rostral periaqueductal gray (Satpute et al., 2013). The periaqueductal gray threat response system is likely co-opted in the pathophysiology of conditions such as panic disorder and generalized anxiety disorder. Blood flow analysis suggests that the inhibitory influence of the cortex over the fight-or-flight mechanisms in the periaqueductal gray is reduced in panic disorder (Del-Ben and Graeff, 2009). Functional MRI has also revealed activation of the human periaqueductal gray in complex emotions such as frustration (Yu et al., 2014), admiration and compassion (Immordino-Yang et al., 2009), in addition to more immediate threat responses (Lindner et al., 2015).
Medial Part of the Emotional Motor System
The medial part of the emotional motor system (Figures 3, 4) consists of descending projections from the reticular formation that are involved in level-setting and modulatory functions (Holstege, 2009). Once again, the vast majority of the research on this subject has been in animals. The caudal third of the locus coeruleus (Sasaki et al., 2008) and the caudal raphe nuclei both send projections downward to the spinal cord, as depicted in Figure 4, and are responsible for descending pain modulation (Renn and Dorsey, 2005). The effect of norepinephrine from the locus coeruleus is mostly antinociceptive, while serotonin from the raphe nuclei can have varying effects depending upon the type of receptor activated (Benarroch, 2008). In rats, it has been shown that the midbrain tectum and the dorsal/lateral periaqueductal gray indirectly produce the analgesia that occurs in fear (Coimbra et al., 2006), through a primarily non-opioid mechanism involving GABAergic and serotonergic neurons (as opposed to the ventrolateral periaqueductal gray that produces a long-lasting opioid mediated analgesia; Gauriau and Bernard, 2002). It is likely that this system of fear suppressing the pain system is still present in humans, allowing us to act and move rapidly in situations of threat (Mobbs et al., 2007).
In addition to nociceptive modifications, the medial part of the emotional motor system is also involved in level-setting for arousal levels and muscle function – studies on rodents and monkeys indicate that this is accomplished through norepinephrine secretion from the locus coeruleus (Aston-Jones and Cohen, 2005; Lang and Davis, 2006) and cholinergic projections from the pedunculopontine tegmental nucleus in the upper pons (Bechara and van der Kooy, 1989; Homs-Ormo et al., 2003). Further detail regarding these important structures is provided in the section below on the Modulatory network.
Summary statement: The Descending network, otherwise referred to here as the emotional motor system, has a lateral part that triggers patterned emotional behaviors, while the medial part is responsible for level-setting in sensory and arousal systems that might be important in emotionally charged situations.
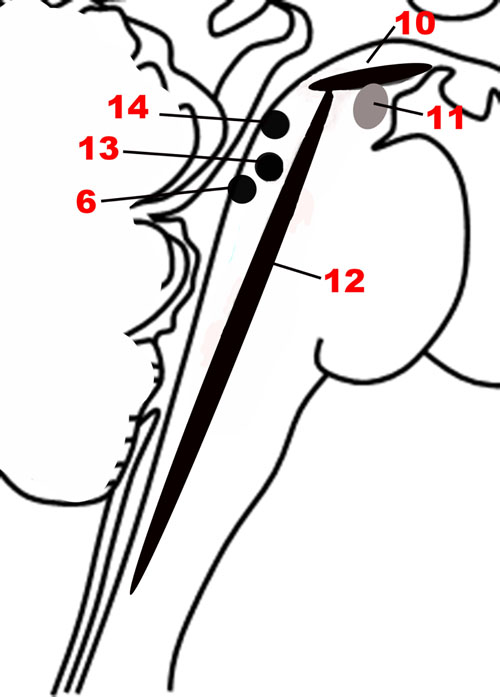
Modulatory Neurotransmitter Network – Valence, Arousal, and Reward
Since a major characteristic of an adaptive emotional behavioral response is flexibility, a network that modulates the autonomic, motor, affective and memory changes brought about by different stimuli is needed. The chief upper brainstem structures involved in this modulation are the neurotransmitter pathways arising from the upper raphe nuclei (serotonergic), the ventral tegmental area-substantia nigra pars compacta complex (dopaminergic), and the upper locus coeruleus (noradrenergic), which project widely throughout the hypothalamus, cortex and other parts of the forebrain. In addition, the laterodorsal and the pedunculopontine tegmental nuclei are sources of cholinergic fibers, which stimulate cortical activation through the thalamus. These structures are depicted in Figures 1, 5. Ascending projections from the brainstem to subcortical and cortical structures communicate the states of brainstem structures to more rostral regions of the nervous system, where these states contribute to affective experience. Since these pathways are involved in arousal and in the maintenance of consciousness (Jones, 2003), they are sometimes called the Ascending Reticular Activating System or Ascending Arousal Network (Moruzzi and Magoun, 1949; Edlow et al., 2012). The following sections on the various pathways that comprise the Modulatory network are in large part descriptions of the Ascending Reticular Activating System, albeit with a focus on how these relate to emotion.
The Valence-Arousal Model of Emotion and Its Critiques
The modulation of affective states by these upper brainstem-based pathways has been expressed through the two domains of valence and arousal. According to the circumplex model of emotions, each basic emotion is postulated to be a combination of these two domains, in differing degrees (Russell, 1980; Zald, 2003; Posner et al., 2009). In humans, valence correlates with pleasantness ratings, heart rate, and facial muscle activity, while arousal correlates with skin conductance, interest ratings and viewing time for stimuli (Lang and Davis, 2006). Both valence and arousal have significant impact on an organism’s relationship with the environment, influencing, for example, the allocation of attention and long term memory formation (Arbib and Fellous, 2004).
Recent work, especially in the neuroimaging literature, has raised questions about whether complex neurological processes like emotions can actually be represented by reducing to dimensions of valence and arousal. Kragel and LaBar (2016), in an interesting review of the nature of brain networks that subserve human emotion, argue that each emotion uniquely correlates with activation of a constellation of cortical and subcortical structures (Kragel and LaBar, 2016), and that the current neuroimaging data do not support the valence-arousal model of emotions. They focused on fMRI studies which have applied novel statistical methods collectively known as multivoxel pattern analysis to identify mappings between mental states and multiple measures of neural activity. The mainstay of earlier neuroimaging research on emotion was univariate pattern analysis, but multivariate analyses have the advantages of higher sensitivity, and the ability to detect counterintuitive relationships because of the lack of reliance on a priori hypotheses. These approaches also have the advantage of overcoming the assumption that dedicated modules or homogeneous neural units subserve each emotion, because they can investigate various neuronal populations at much larger spatial scales.
Kragel and LaBar (2016) suggest that while the use of machine learning approaches to large neuroimaging datasets is likely to expand in the near future, it might be premature to draw conclusions about neural substrates underlying each emotion, because the current studies using multivariate analyses have not all been consistent with one another. These differences may be coming from technical variations in the methods used to induce and assess the emotion and associated neural activations, but might also represent fundamental variations in the circuitry employed in different individuals, or even a lack of emotional “essences” that can be studied in a standardized manner across people and cultures. While this is a valid critique, we believe that the older valence-arousal classification still holds value in furthering our understanding of brainstem contributions to emotions and especially to basic emotions shared with intelligent animals. This debate may eventually be resolved with technical advances in functional neuroimaging and multidisciplinary approaches to studying emotional experiences (Immordino-Yang and Yang, 2017, in press).
Conclusion and Future Directions
The brainstem contains several structures that are likely of critical importance in the generation and experience of emotion. Most prior research on human emotion has focused on cortical mechanisms, largely because of the complexity of the brainstem coupled with the difficulty of analyzing brainstem functioning using current technologies. We have provided a conceptual overview of how tegmental structures of the brainstem are involved in emotion-related processes. Future research on the structural and functional connectivity of the human brainstem is needed to further understand its role in emotion. Such work will undoubtedly contribute to a more enriched and nuanced understanding of the neurobiology of human emotion in psychology and in affective neuroscience.